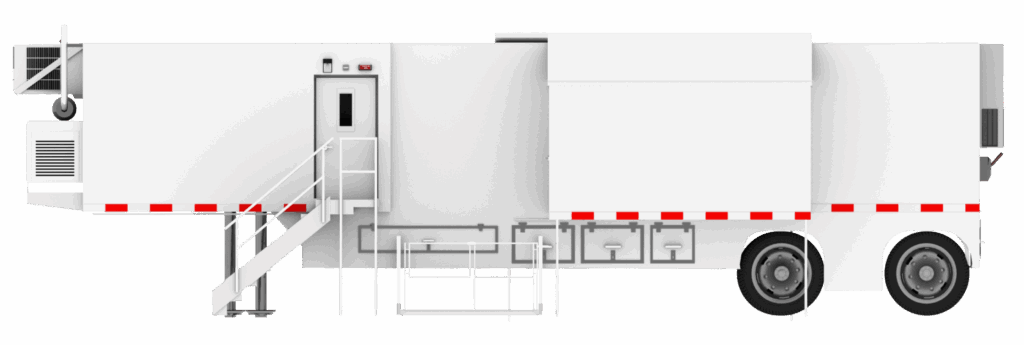
Healthcare facilities that perform sterile or hazardous drug compounding must clearly understand the difference between USP <797> and USP <800> and how each regulation impacts cleanroom design. While both standards regulate compounding pharmacy environments, they serve different purposes and require different cleanroom strategies.
For hospitals, health systems, oncology centers, and outsourcing facilities, understanding USP <797> and USP <800> requirements is critical before building, renovating, or deploying a cleanroom solution, especially when evaluating permanent versus mobile cleanroom options.
Understanding USP <797> Requirements
USP <797> governs sterile compounding. Its primary purpose is to protect patients by ensuring compounded sterile preparations are free from microbial contamination and particulates.
Cleanrooms that house USP <797> operations must meet strict environmental standards, including ISO air classification, air changes per hour, pressure differentials, temperature and humidity control, and environmental monitoring protocols. In most non-hazardous sterile compounding applications, the cleanroom operates under positive pressure. This airflow strategy cascades clean air, pushing it outward, preventing contaminants from entering the sterile compounding environment.
A compliant USP <797> cleanroom typically includes an ISO 8 ante room, an ISO 7 buffer room, and an ISO 5 primary engineering control, such as a laminar flow hood or biological safety cabinet. These elements work together to maintain sterility assurance and product integrity.
Failure to meet USP <797> cleanroom standards can result in regulatory citations, accreditation issues, and potential patient safety risks.
Understanding USP <800> Requirements
USP <800> focuses on the safe handling of hazardous drugs. While USP <797> is centered on patient protection through sterility, USP <800> focuses on personnel and environmental protection through containment.
A USP <800> cleanroom must be designed to prevent exposure to hazardous drugs, including chemotherapy agents and other medications that pose occupational risks through exposure. The defining feature of a USP <800> cleanroom is negative pressure. Unlike the positive pressure used in most USP <797> cleanroom environments, a USP <800> cleanroom must pull air inward to ensure hazardous particles do not escape into adjacent areas.
USP <800> cleanroom environments also include externally vented engineering controls, defined containment secondary engineering controls, and strict decontamination procedures. When sterile hazardous drugs are compounded, the cleanroom must comply with both USP <797> and USP <800> simultaneously. This dual compliance requirement makes proper cleanroom design especially critical.
USP <797> vs. USP <800>: The Core Difference
At a technical level, the difference between USP <797> and USP <800> cleanroom environments comes down to airflow and protection priorities.
A USP <797> cleanroom typically uses positive pressure to protect the sterile product. A USP <800> cleanroom uses negative pressure to contain and eliminate hazardous drug particles and protect staff. When both sterile and hazardous compounding occur within the same facility, pressure cascades must be engineered carefully to maintain compliance with both standards.
Improper cleanroom pressure relationships are one of the most common inspection deficiencies.
The Challenge of Traditional Cleanroom Construction
Building or retrofitting a permanent USP <797> or USP <800> cleanroom can require a significant capital investment and typically have extended timelines. Design coordination, mechanical systems integration, certification, and validation can take months. For facilities undergoing renovation or responding to inspection findings, this delay can disrupt compounding operations and impact patient care.
Additionally, regulatory expectations continue to evolve. A traditional cleanroom build may require modifications over time as interpretations of USP <797> and USP <800> standards are refined.
This is where mobile cleanroom solutions provide a strategic advantage.
The Benefits of a Mobile Cleanroom for USP <797> and USP <800> Compliance
A mobile cleanroom offers a flexible and rapidly deployable alternative to conventional construction. A properly engineered mobile cleanroom can deliver a fully compliant USP <797> environment, a USP <800> hazardous drug containment space, or a dual-compliant configuration—without the long timelines of permanent construction.
Modular Devices’ mobile cleanroom fleet is specifically designed to meet USP <797> and USP <800> requirements. Each mobile cleanroom is engineered with validated ISO classifications, appropriate pressure differentials, and integrated environmental monitoring systems. For hazardous drug compounding, negative pressure and external exhaust systems are incorporated to satisfy USP <800> cleanroom standards.
Mobile cleanrooms can support facilities during renovations, expand their sterile compounding capacity, or provide rapid remediation after regulatory events. Because they are pre-engineered, preconstructed, and performance tested, deployment timelines are significantly shorter than traditional builds—often in under two weeks.
Integrated Modular Cleanroom Solutions for Dual Compliance
For facilities that require both USP <797> and USP <800> compliance, an integrated modular cleanroom solution offers a powerful advantage. Our compounding pharmacy cleanroom lease fleet allows both a positive-pressure USP <797> cleanroom and a negative pressure USP <800> cleanroom to operate within the same controlled structure, but in clearly segregated, pressure-appropriate zones.
This integrated approach ensures sterility for non-hazardous sterile compounding while maintaining full hazardous drug containment where required. Our units are the only mobile solutions on the market to also contain a dedicated hazardous unpack and storage room for USP <800> materials. By housing both USP <797> and USP <800> cleanroom environments within a single modular system, facilities gain operational efficiency, regulatory confidence, and scalability.
Whether deployed as a permanent commercial modular installation or as a mobile cleanroom solution, this dual-compliant design minimizes construction risk, reduces downtime, and provides flexibility as regulatory standards evolve.
Choosing the Right Cleanroom Strategy
USP <797> and USP <800> are not interchangeable standards. Each requires a distinct cleanroom strategy, airflow design, and engineering approach. Facilities that compound sterile preparations, hazardous drugs, or both must ensure their cleanroom environment aligns precisely with regulatory expectations.
If your organization is planning a cleanroom build, renovation, expansion, or upgrade, a mobile or modular USP <797>/<800> cleanroom solution can provide a faster, lower-risk path to compliance. With the right design and deployment strategy, you can meet regulatory requirements confidently, while maintaining uninterrupted patient care and operational continuity. Learn more about how Modular Devices Cleanrooms can deliver the space you need when you need it.