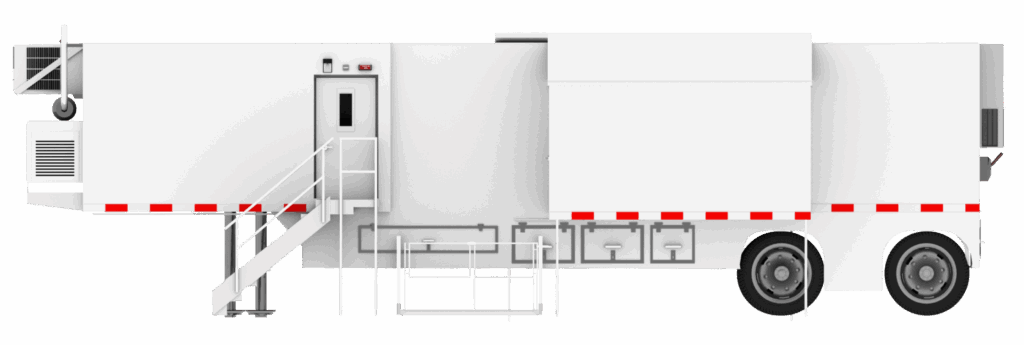
As cardiovascular volumes continue to rise and care increasingly shifts toward outpatient and decentralized settings, hospitals and cardiology centers are rethinking how they plan, expand, and protect cath lab capacity. One solution gaining widespread adoption is the mobile cath lab, a flexible, fully functional cardiac catheterization environment delivered where and when it’s needed.
Mobile cath labs are no longer viewed as emergency-only assets. Today, they play a strategic role in long-term growth planning, operational continuity, and access expansion across hospitals, health systems, and cardiovascular service lines.
This article breaks down what a mobile cath lab is, how it’s used, and what providers should consider when evaluating mobile cardiac lab solutions.
What Is a Mobile Cath Lab?
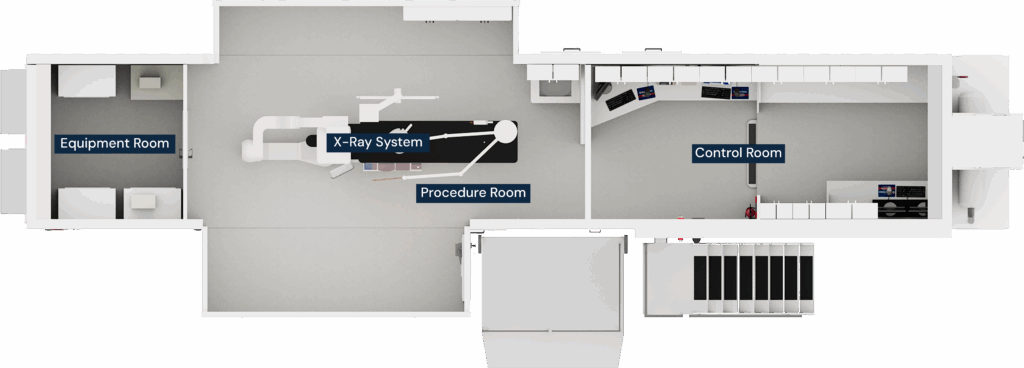
A mobile cath lab is a fully equipped cardiac catheterization lab housed in a trailer-based or modular structure that can be delivered to a hospital, health system, or cardiovascular center on a temporary or semi-permanent basis. Once installed, the lab functions as a complete procedural environment, supporting a wide range of diagnostic and interventional cardiology procedures.
Unlike fixed cath labs that require permanent construction, mobile cath labs allow providers to add capacity without long construction timelines, major capital investments, or operational disruption.
Common Use Cases
Mobile cath labs are used in a variety of clinical and operational scenarios, including:
- Temporary capacity expansion during periods of increased procedure volume
- Overflow support when existing cath labs are operating at or near full utilization
- Renovation or equipment replacement coverage to maintain uptime during construction
- Outreach programs supporting satellite campuses or underserved regions
- Disaster recovery and emergency preparedness, ensuring continuity of cardiovascular services
In each case, the goal is the same: preserve access to care while maintaining procedural quality, safety, and compliance.
Typical Mobile Cath Lab Configurations
Mobile cardiac lab solutions are available in several formats, allowing providers to select the configuration that best fits their space, volume, and long-term planning needs.
Trailer-Based Mobile Cath Labs
Trailer-based mobile cath labs are the most widely recognized configuration. These units arrive pre-built and fully equipped, including:
- Procedure room with a fixed cardiac imaging system
- Control room and equipment space
- Patient prep and recovery areas (depending on layout)
- Integrated shielding, HVAC, power distribution, and medical gas systems
Once delivered and connected to site utilities, the lab can be operational quickly, often in days.
Modular Cath Labs
Modular cath labs are constructed off-site and installed on-site as semi-permanent structures. While still faster than traditional construction, modular solutions are typically used when longer deployment durations or integration into a broader facility expansion are required.
Hybrid Approaches
Some providers deploy mobile labs as interim solutions while planning permanent expansions or transitioning services to new campuses. In these cases, mobile and modular solutions work together as part of a phased growth strategy.
How Mobile Cath Labs Compare to Fixed Cath Labs
From a clinical perspective, mobile cath labs are designed to mirror the functionality of fixed cath labs as closely as possible. The primary differences lie not in clinical capability, but in deployment speed, financial structure, and operational flexibility, including how capacity is leased, scaled, and timed to demand.
Key Differences:
- Speed to Deployment
Mobile cath labs can be delivered and installed significantly faster than permanent construction projects, allowing providers to respond quickly to rising procedure volumes, physician recruitment, or unexpected downtime.
- Capital vs. Operating Expense
Unlike fixed cath labs, which typically require substantial upfront capital investment, mobile cath labs are most often deployed through flexible leasing arrangements. This structure allows organizations to preserve capital, align costs with actual utilization, and avoid long-term financial commitments while volumes stabilize or grow.
- Flexible Leasing & Scalability
Mobile cath lab leases can be structured for short-term coverage, multi-year use, or extensions as needs evolve. This flexibility enables hospitals and cardiology centers to scale capacity up or down based on real-world demand—without locking into permanent infrastructure decisions before volumes, reimbursement, or strategic direction are fully defined.
- Operational Flexibility
Mobile labs can be relocated, extended, or removed as service line needs change. This makes them well-suited for temporary expansions, bridge solutions during construction, or phased growth strategies.
- Continuity of Care
Mobile cath labs allow cardiovascular programs to remain operational during renovations, equipment upgrades, or facility expansions—helping avoid canceled cases, lost revenue, and disruptions to patient access.
For many providers, mobile cath labs are not a replacement for permanent labs, but a strategic complement—supporting growth while managing financial risk, preserving flexibility, and maintaining confidence in long-term planning.
Regulatory and Accreditation Considerations
Any cath lab—mobile or fixed—must meet strict regulatory and accreditation requirements. Reputable mobile cath lab providers design their solutions to align with current standards and support hospitals throughout the compliance process.
Key considerations include:
- Centers for Medicare & Medicaid Services (CMS) requirements for cardiac procedures, reimbursement eligibility, and ASC alignment, including compliance with the ASC Covered Procedures List criteria
- The Joint Commission accreditation standards related to patient safety, infection control, facility design, and survey readiness for mobile clinical environments
- DNV Healthcare requirements for quality management systems, clinical governance, and continuous compliance
- State-specific licensure, health department, and accreditation considerations, which may vary by jurisdiction for mobile or temporary healthcare facilities, require proactive coordination during planning and deployment
Providers should ensure that mobile cath lab solutions are engineered, installed, and supported in a way that aligns with these regulatory frameworks, including documentation, inspections, and ongoing operational support.
Why Providers Are Increasingly Turning to Mobile Cath Labs
The growing adoption of mobile cath labs reflects broader shifts in healthcare delivery:
- Rising cardiovascular procedure volumes
- Pressure to reduce wait times and improve access
- Capital constraints and reimbursement complexity
- Demand for faster, lower-risk capacity expansion
Mobile cath labs offer a practical path forward—allowing hospitals and cardiology centers to respond quickly to demand while maintaining high clinical and operational standards.
Take the Next Step
Mobile cath labs are no longer just stopgap solutions. For many providers, they are a strategic tool for protecting access, supporting growth, and maintaining continuity of care.
Contact us to explore configurations, planning considerations, and real-world deployment scenarios that can help you determine whether a mobile cath lab is right for your organization.